The AskBio AAV technology platform
Since our founding over 20 years ago and with more than 40 years of pioneering AAV research, our vision has always extended beyond therapeutic development. Making genetic medicines that appropriately target the right cells, and provide benefit to patients in a safe manner is vital to improving patient lives, and the science behind identifying and treating genetic disorders has greatly evolved.
Equally important is helping to ensure that patients in need have access to genetic medicine. It is why we consider our AAV technology platform one of our greatest innovations. Its components are shaping how we scale gene therapy manufacturing and lower production cost.
It is this uncompromised vision that propelled the emergence of our gene therapy technology platform.
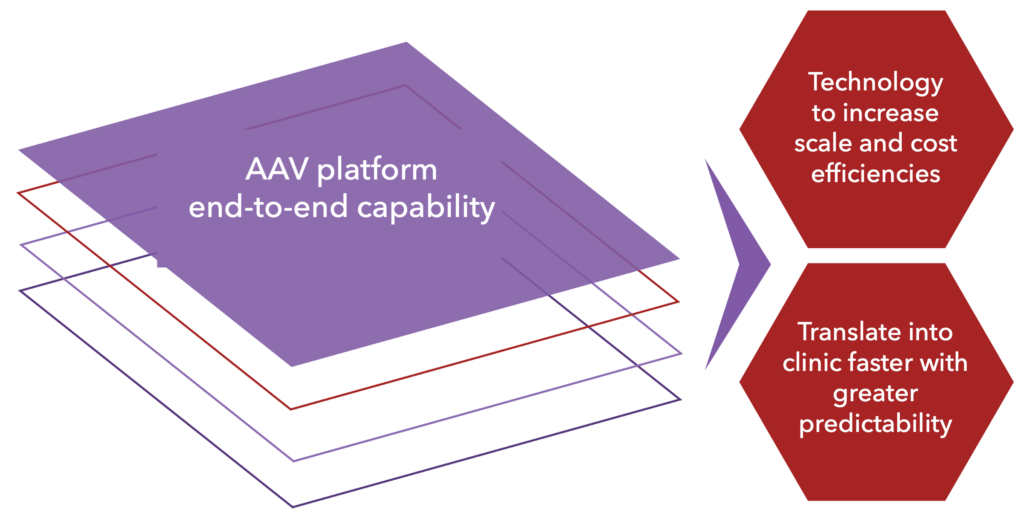
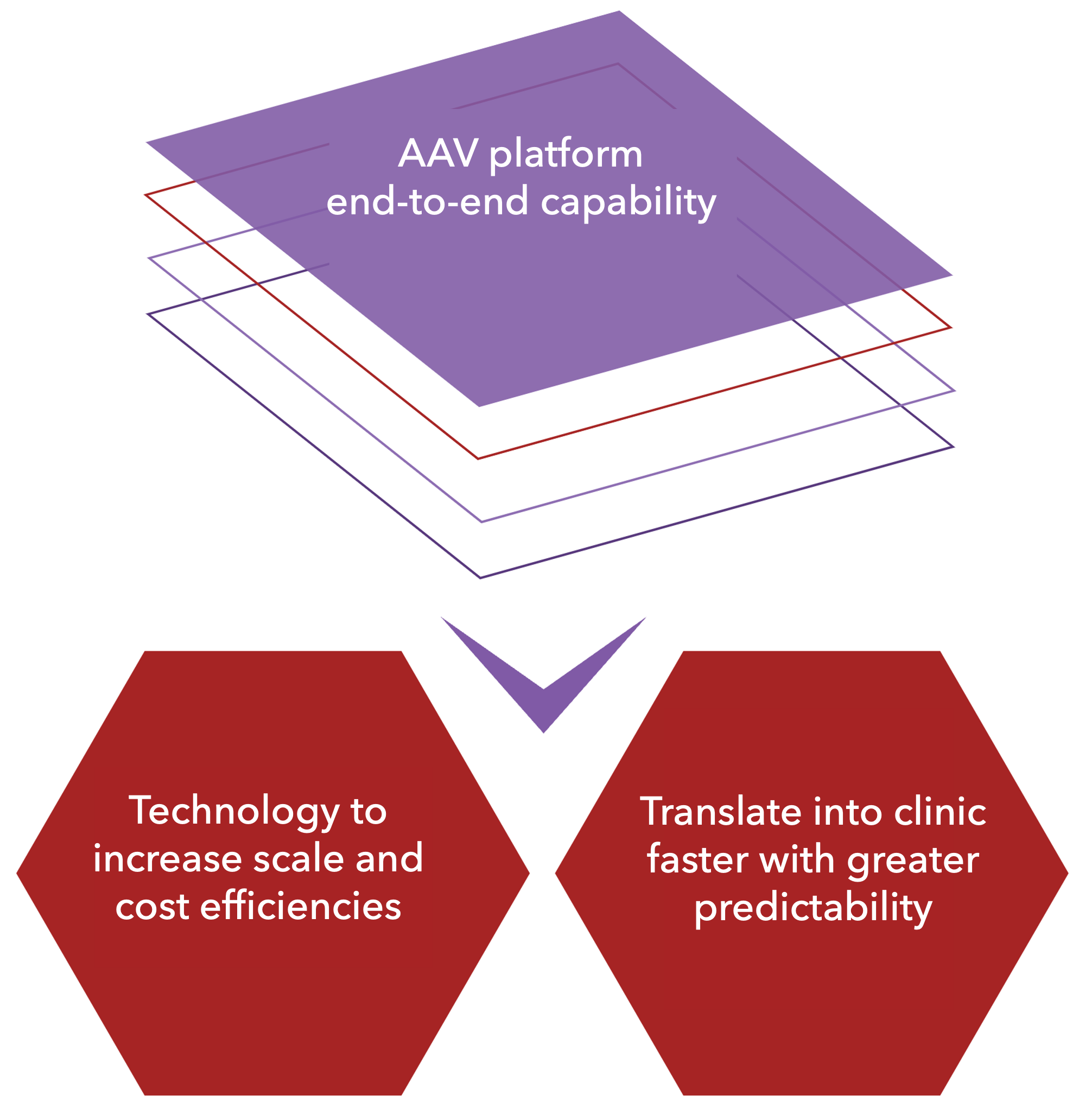
AAVPlatform proven end-to-end capability results in technology to increase scale and lower costs of goods as well as translate into clinic faster, safer and with greater predictability.
Three essential components of the AskBio AAV platform
Capsids
A vast range of capsid pool designed to match a variety of tropism and maximize transduction
Promoters
An expression cassette with gene control, regulation and inducible technology
Production
Pro10™ is a high-yield suspension cell line that increases overall therapeutic output and lowers production cost