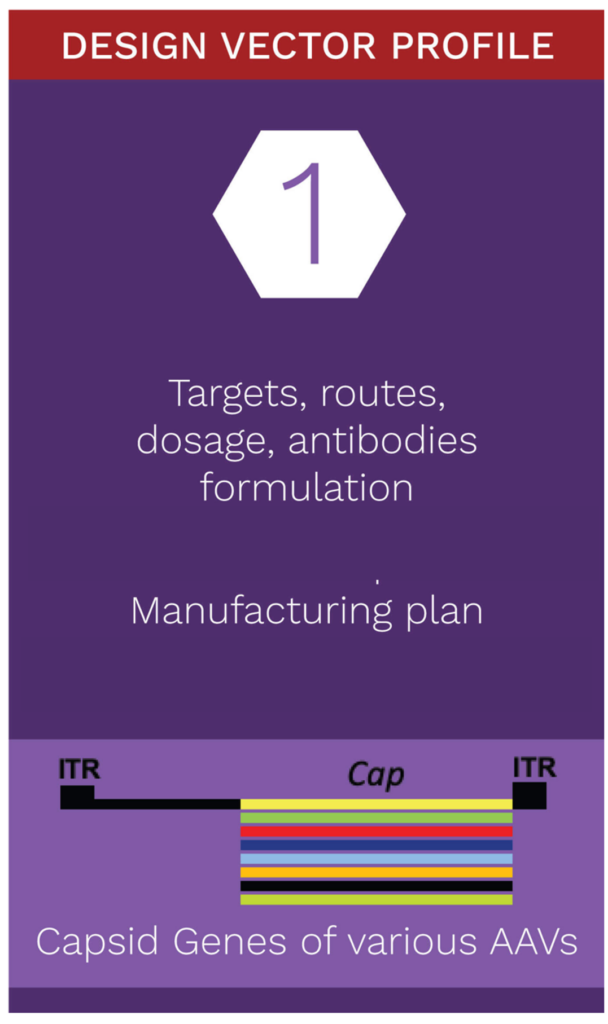
Capsid Development
Naturally occurring serotypes have long enabled delivery of AAV gene therapeutics. However, precise tissue targeting without off-target implications important for advancing therapeutic performance.
Rational design for chimeric capsids with a range of possibilities
A critical component of our gene therapy technology platform is the application of bioinformatics, crystal structure analytics, in vivo/in vitro modeling, and genome sequencing techniques. This allows us to alter the natural tropism range of chimeric AAV vectors.
Our unique rational design process expands the functional capability of proteins from naturally occurring serotypes, as well as proteins from other capsid sequences.



A new generation of chimeric vectors
Chimeric AAV capsids enable efficient gene delivery for a precise tissue target while de-targeting tissues that normally accumulate high levels of AAV.
- Systemic administration for reliable Chimeric vectors help deliver the product to specific cells in the body and can be more accurate than non-chimeric vectors
- Potential to specifically de-target organs, such as the liver
- Systematic approach for mixing and matching natural serotype proteins and any capsids for efficient transduction
- A screening process combines bioinformatics, applied clinical performance and animal study data
Novel capsids may maximize transduction
Heparin and heparan sulfate binding chimeric vectors
This family of chimeric capsids contains modified receptor-recognition sites that change the cell or tissue tropism of the capsids, shown to mediate modified transduction of target cells in different types of tissue.
Inner Loop chimeric vectors
Chimeric capsids with modified inner-loop regions have shown novel biodistribution and transduction patterns after systemic administration. Primarily for heart and skeletal muscles, inner-loop vectors can increase the therapeutic index with some inner-loop variants showing 40x reduction of liver transduction while retaining high-level tropism for other target tissues (Asokan A., 2010; Shen S. 2013). These properties have encouraged the use of inner-loop mutants in clinical programs targeting cardiac muscle.
Dual Glycan receptor chimeric vectors
In addition to demonstrating the modularity of glycan receptor footprints on viruses, our approach provides design parameters to upgrade the current AAV vector toolkit for clinical gene therapy. New AAV capsids can be evolved to recognize different host glycans through mutagenesis and experimental adaptation. These AAV capsid mutations affect viral binding to cells with carbohydrate receptors, demonstrating modularity of glycan receptor footprints on viruses. This exploits two distinct glycan receptors for cell entry and results in a novel class of adeno-associated viruses (AAV).
Dual glycan binding AAV
Prototyping
A prototypical dual glycan binding AAV strain was engineered by “grafting” a galactose (Gal) binding footprint from AAV serotype 9 onto the heparan sulfate (HS) binding AAV serotype 2.
The resulting chimera, AAV2G9, interchangeably exploits Gal and HS as evidenced by competitive inhibition assays with lectins and glycans (Shen S., 2013). AAV2G9 mediates rapid onset and sustained, higher transgene expression in vivo compared to parental AAV serotypes.
This demonstrates the modularity of glycan receptor footprints on viruses and design parameters to upgrade AskBio’s AAV vector toolkit available for investigational gene therapy.
Are you our next great collaborator?
Harness the power of our technology, scientific expertise, and clinical process experience to help bring discoveries to life.




