Clinical Trial
Congestive Heart Failure (CHF)
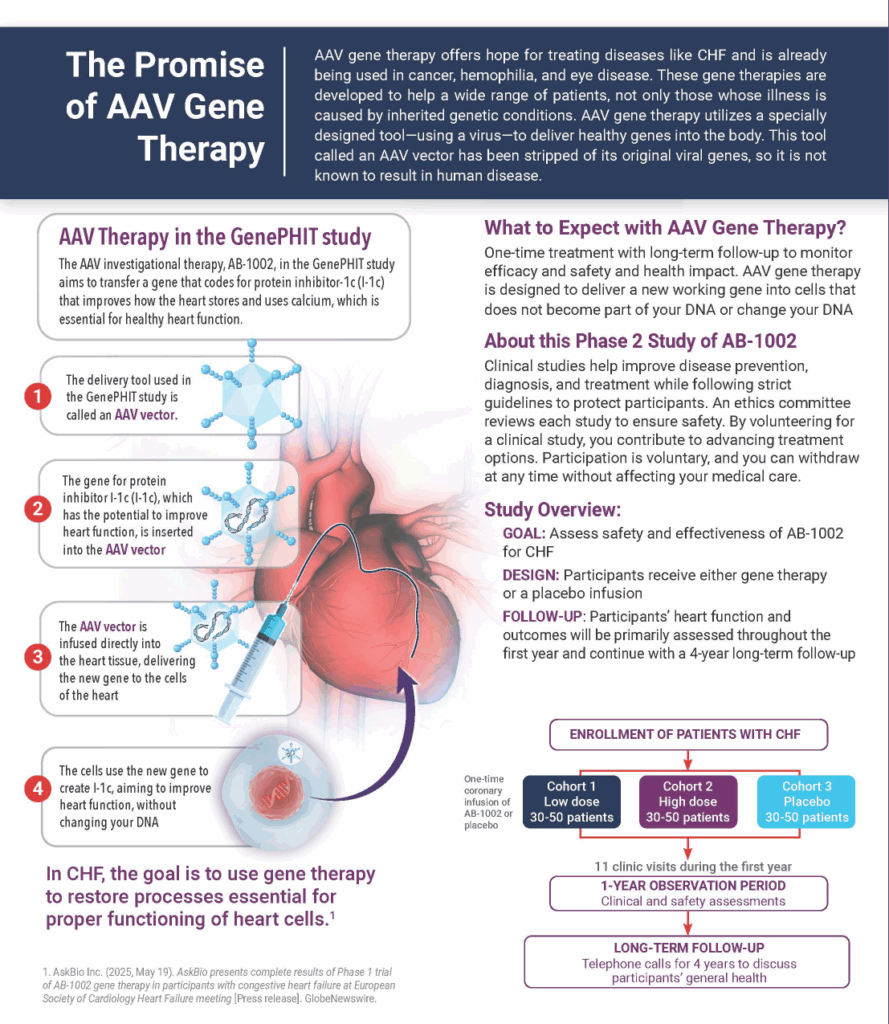
GenePHIT (Gene PHosphatase Inhibition Therapy) is a Phase 2 adaptive, double-blinded, placebo-controlled, randomized, global multi-center trial to evaluate the safety and efficacy of the one-time administration of AB-1002, an investigational gene therapy that aims to activate a key protein to stop the action of type 1 protein phosphatase (PP1), a substance that plays an important role in the development of heart failure, in people with CHF.
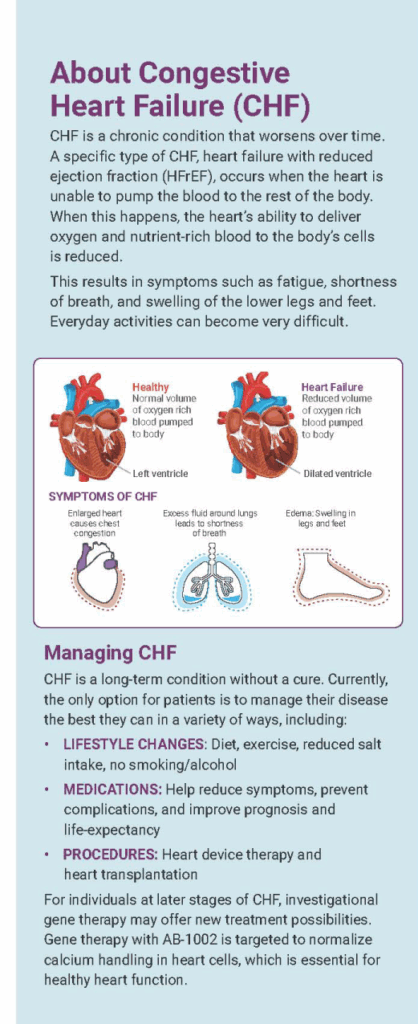
What is CHF?
CHF is a chronic condition that gets worse over time. It can result from the weakening or stiffening of the heart muscle, or from increased oxygen demand by body tissues beyond the heart’s capability. CHF happens when the heart is unable to supply enough blood and oxygen to the body.
Patients with CHF may experience a range of debilitating symptoms, including:
- Shortness of breath
- Coughing/wheezing
- Weight gain
- Swollen ankles, legs or abdomen
- Tiredness/fatigue
- Dizziness
- Rapid heart rate
CHF is a chronic disease that severely impacts quality of life; if you think you have CHF, please talk to a medical professional.
About the GenePHIT Trial
GenePHIT is a Phase 2 adaptive, double-blinded, placebo-controlled, randomized, multi-center trial to evaluate the safety and efficacy of the one-time administration of AB-1002, via antegrade intracoronary artery infusion, in males and females age >18 years with cardiomyopathy and New York Heart Association (NYHA) Class III heart failure symptoms.4 Subjects are randomized into one of three treatment groups in a 1:1:1 fashion to either low dose, high dose, or placebo. Primary outcome measures include cardiovascular related death and change from baseline in NYHA classification, left ventricular ejection fraction (LVEF), and Six Minute Walk Test (6MWT).4
Learn more about the this study at clinicaltrials.gov (NCT#05598333).
AB-1002 is an investigational therapy and has not been approved by the United States Food & Drug Administration (FDA) or any other healthcare authority for commercial use.
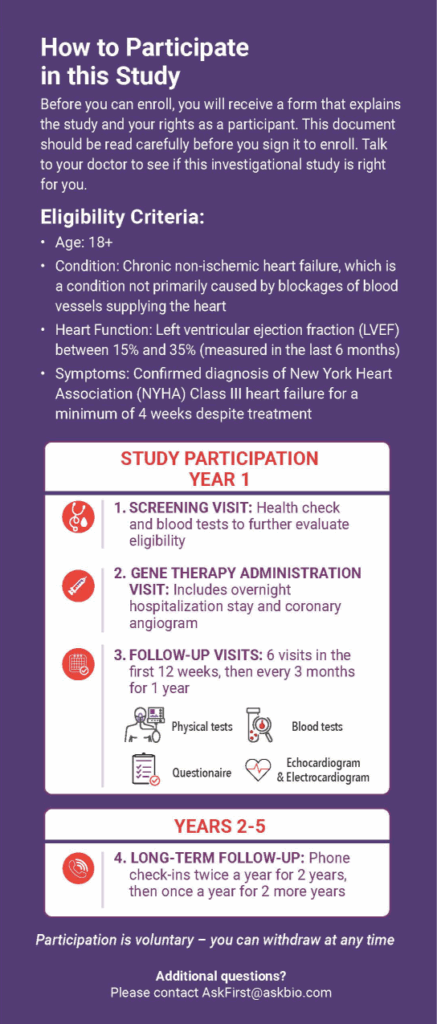
Clinical trial activities are outlined below:
Screening Period
Used to determine eligibility.
Gene Therapy Administration
Administration of one-time infusion of AB-1002 by cardiac catheterization and infusion into the arteries of the heart.
Health Assessments
Occur approximately weekly to twice a month for the first 3 months following administration, then every three months for the remainder of the first year.
Long-Term Follow-Up Period
After the first year of participation post-administration, follow-up visits will occur every 6 months for 2 additional years.
Are You Eligible?
All clinical trials have specific eligibility criteria that patients must meet to participate. These are called inclusion and exclusion criteria and are related to a person’s health when they enter the clinical trial.
Patients may be eligible if they:
- Are over 18 years of age
- Have chronic non-ischemic cardiomyopathy
- Have New York Heart Association (NYHA) Class III heart failure symptoms and have been medically stable for at least four weeks
Patients are not eligible if they:
- Have chronic ischemic cardiomyopathy
- Have received intravenous (IV) inotropic therapy, intra-aortic balloon pump (IABP) or percutaneous cardiac assist device therapy within 30 days prior to enrollment
- Have restrictive cardiomyopathy, obstructive cardiomyopathy, pericardial disease, amyloidosis, infiltrative cardiomyopathy, uncorrected thyroid disease or dyskinetic left ventricular (LV) aneurysm
- Have had cardiac surgery or percutaneous coronary intervention (PCI) within 30 days prior to enrollment
- Have third degree heart block
- Have had a clinically significant myocardial infarction (MI) within 6 months prior to enrollment
The GenePHIT Study
To find out if you are eligible for this clinical trial, you should talk to your doctor, or to find out more about this study, please visit clinicaltrials.gov.